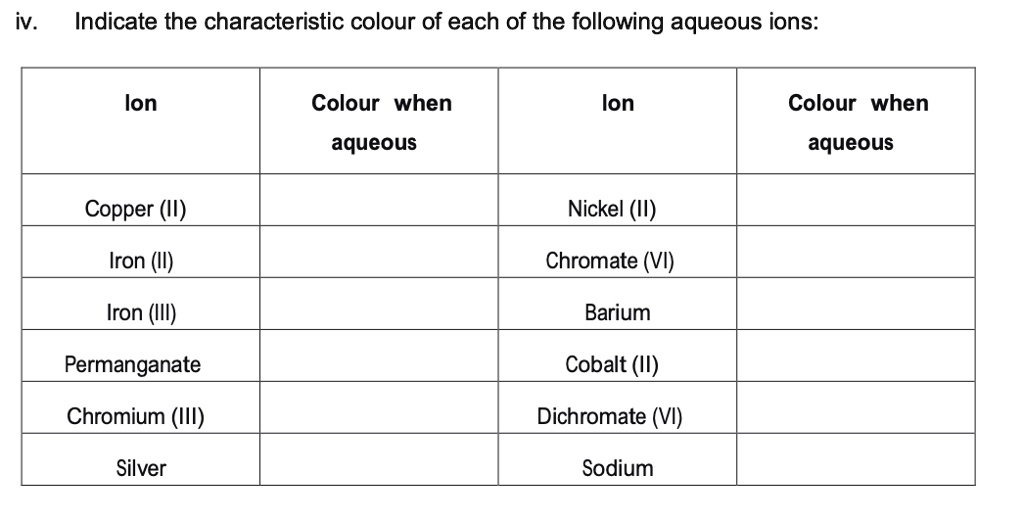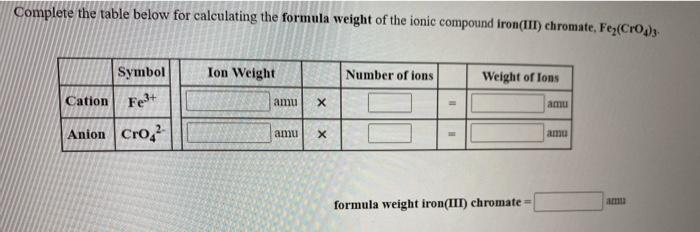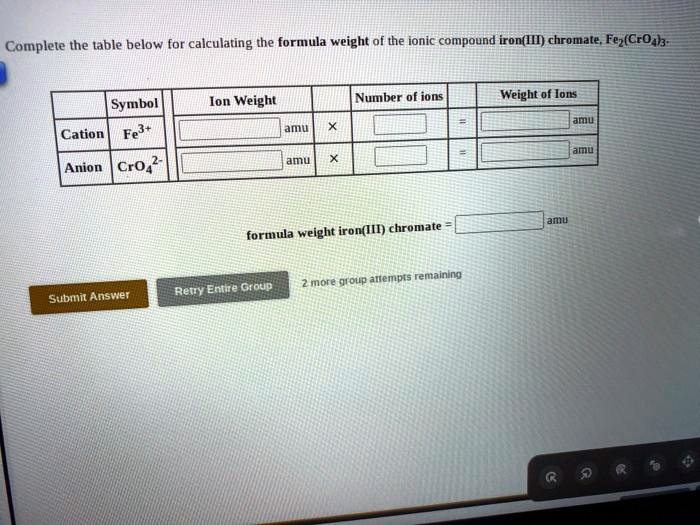
SOLVED: Complete the table below for calculating the formula weight of the ionic compound iron(II) chromate; Fe(CrO3) Ion Weight Number of ions Weight of ions Symbol Cation Fe2+ ? amu Anion CrO4^2- ?

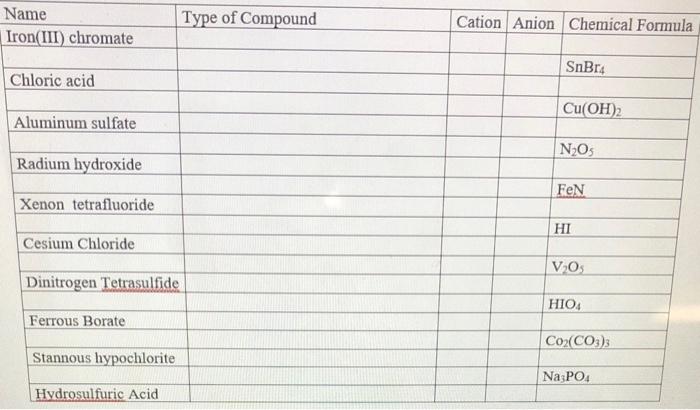
SOLVED: Write the formula for each ionic compound. a. copper(II) chloride b. copper(I) iodate c. lead(II) chromate d. calcium fluoride e. potassium hydroxide f. iron(II) phosphate

SOLVED: To determine the salt formed during a neutralization reaction, combine the anion formed by the dissociation of the acid and the cation formed by the dissociation of the base in a

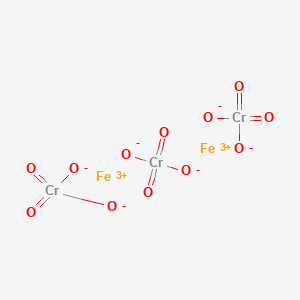
SOLVED:Many chromate (CrO4^2-) salts are insoluble, and most have brilliant colors that have led to their being used as pigments. Write balanced net ionic equations for the reactions of Cu^2+, Co^3+, Ba^2+,
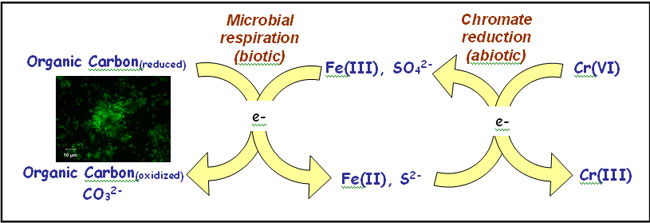
Fate and Stability of Cr Following Reduction by Microbially Generated Fe(II) | Stanford Synchrotron Radiation Lightsource

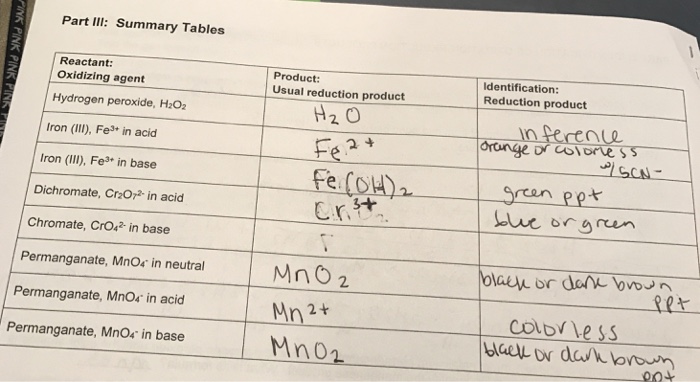
Station A 1)Name the following compounds: CaBr 2 calcium bromide RbOHrubidium hydroxide Li 2 CrO 4 lithium chromate 2)Write the formula for the following. - ppt download