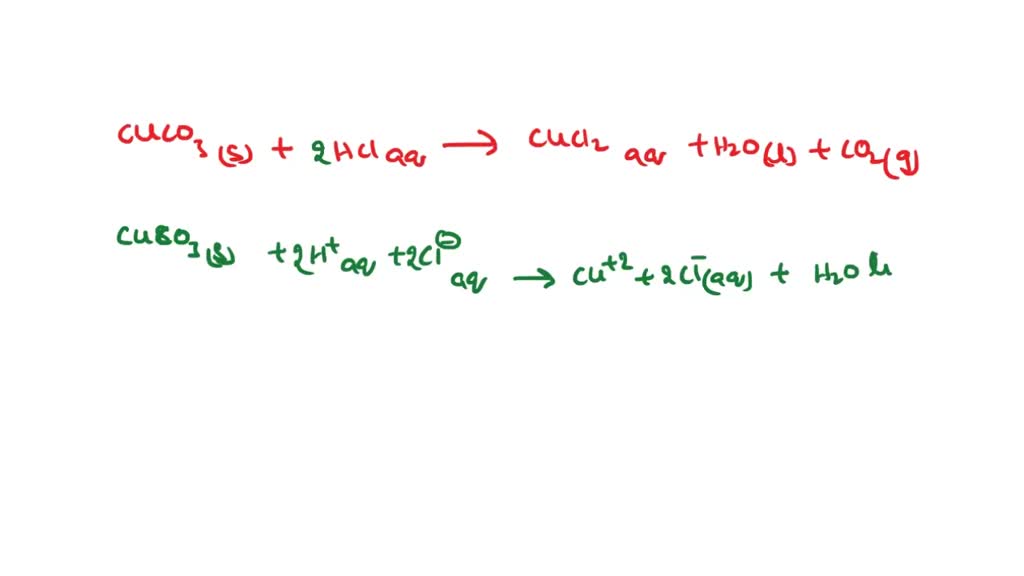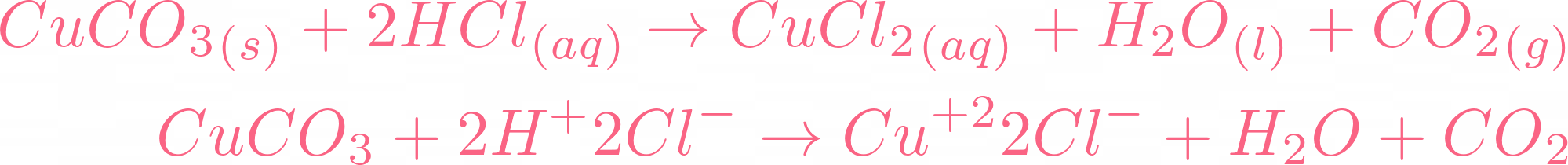SOLVED: Reagents: Original Appearance: CuCO3: The color of this solid is green. HCl (aq): The color of this solution is clear. The Reaction between the two solutions produces a gas. The Molecular
i) Dilute nitric acid and Copper carbonate. (ii) Concentrated hydrochloric acid and Potassium permanganate solution. - Sarthaks eConnect | Largest Online Education Community
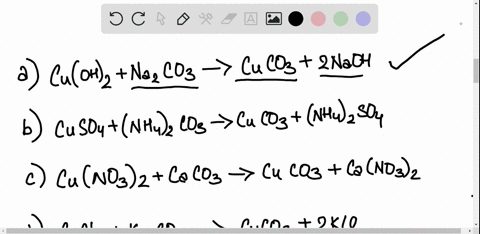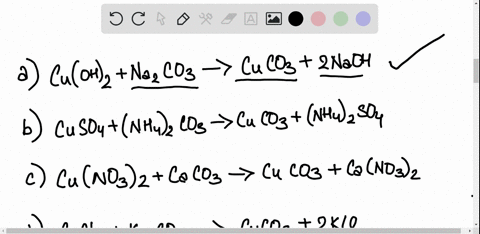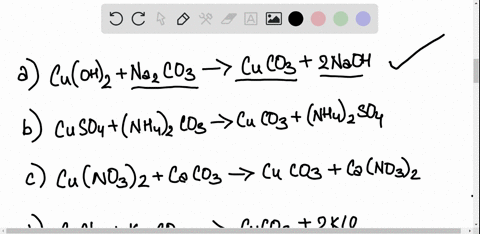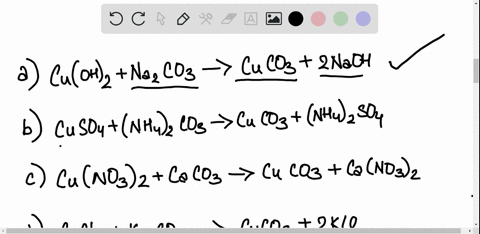
SOLVED: Copper carbonate (CuCO3) reacts with hydrochloric acid (HCl) according to this equation: CuCO3(s) + 2HCl(aq) â†' CuCl2(aq) + H2O(l) + CO2(g). Which statement correctly describes the substances in this reaction? A.