Chapter 6 Thermochemistry. 6.1 Chemical Hand Warmers 6.2 The Nature of Energy: Key Definitions 6.3 The First Law of Thermodynamics: There is no Free Lunch. - ppt download

Chapter 6 Thermochemistry. 6.1 Chemical Hand Warmers 6.2 The Nature of Energy: Key Definitions 6.3 The First Law of Thermodynamics: There is no Free Lunch. - ppt download
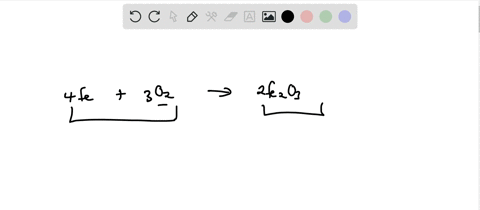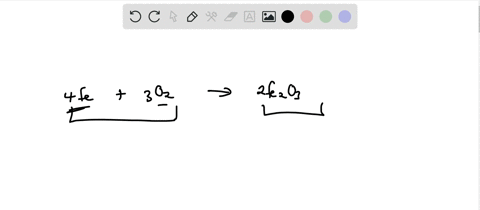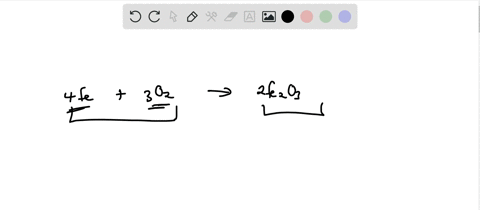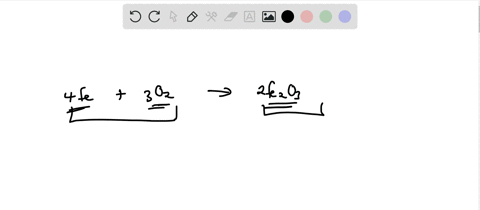
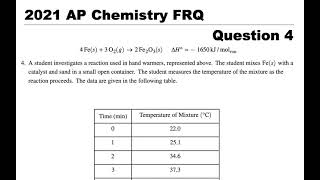
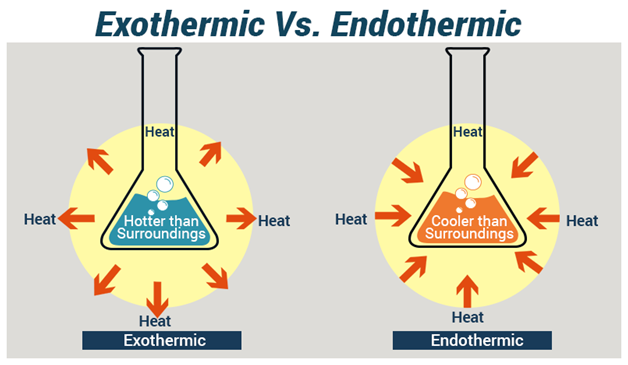
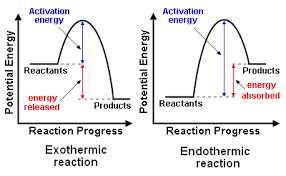
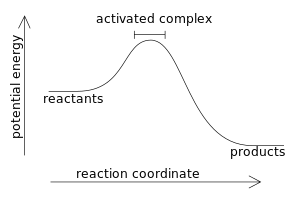
SOLVED: Write the formation equation for iron(III) oxide. Chemical hand warmers popular with skiers and snowboarders produce heat when they are removed from their airtight plastic wrappers. They utilize the oxidation of iron to form iron(III) oxide according ...
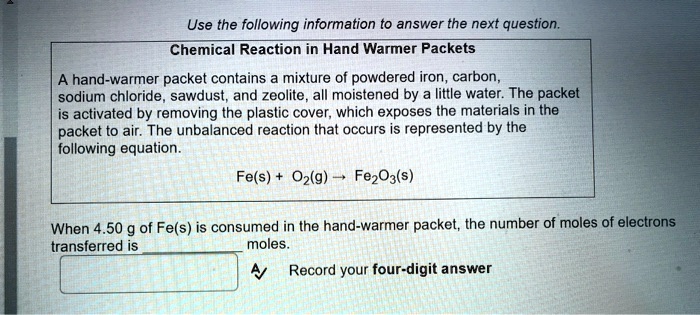
SOLVED: Chemical Reaction in Hand Warmer Packets A hand-warmer packet contains a mixture of powdered iron, carbon, sodium chloride, sawdust, and zeolite, all moistened by a little water. The packet is activated